


Reveal Clinical translates teams' trial data into predictive models.






Impact across the entire clinical and commercial lifecycle
through optimized patient selection and enrichment
for critical efficacy and safety data
through broader reimbursement coverage supported by subgroup-level evidence
integrate internal trial data with published literature and real-world evidence
construct and validate disease progression models
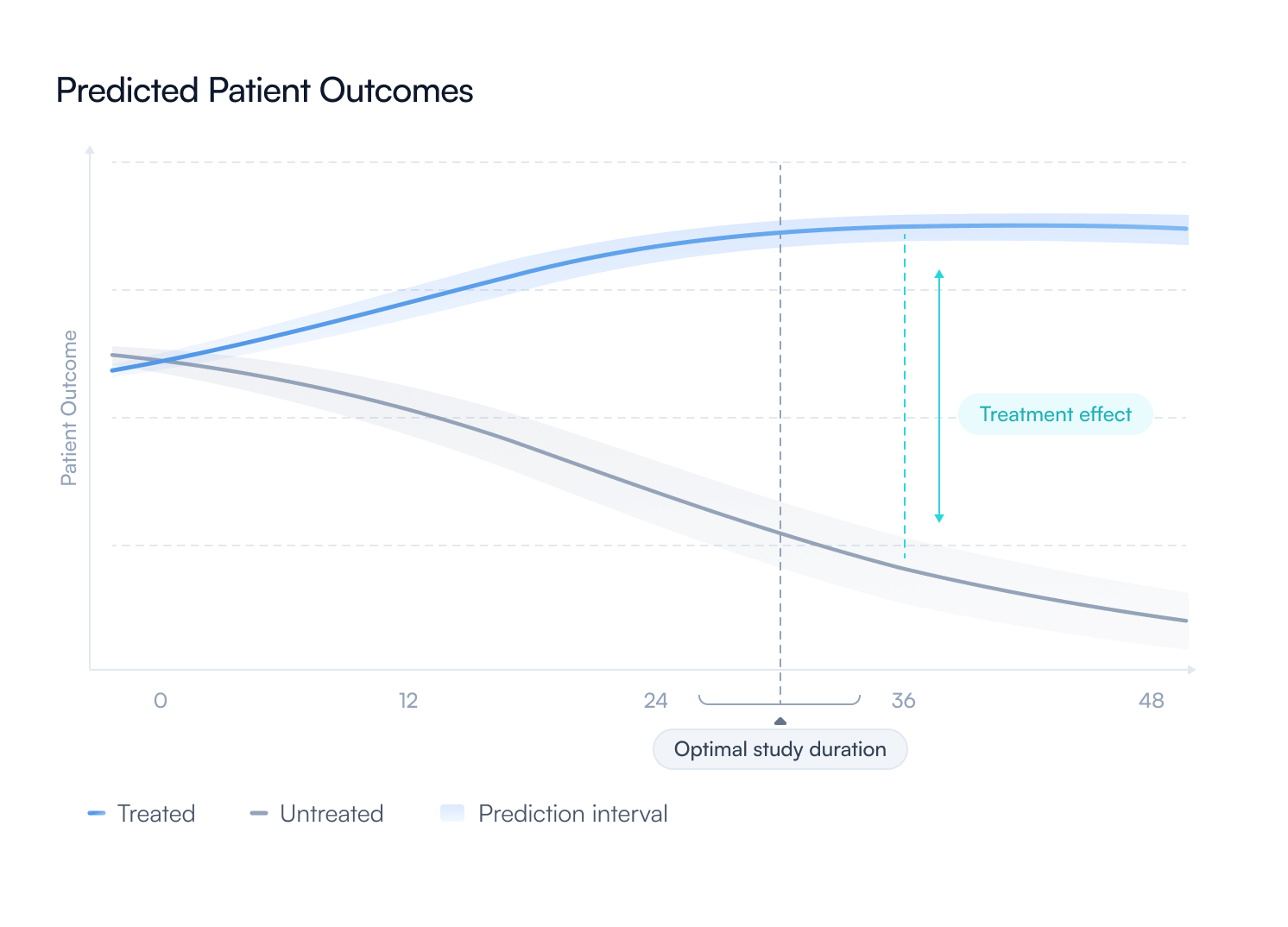
simulate trial designs and project clinical outcomes
Reveal Clinical is not a replacement for your EDC system. Instead, we sit on top of your existing EDC system and connect out of the box with popular EDC systems such as Medidata Rave and Merative Zelta.
Model-Informed Drug Development is a quantitative approach to drug development, leveraging mathematical and statistical models that represent how drugs and disease affect patients. The most widely utilized MIDD technique is pharmacokinetic (PK) modeling which is universally used to determine optimal drug dosage levels. In addition to PK, we also leverage techniques such as Disease Progression Modeling (DPM), Quantitative Systems Pharmacology (QSP) and Model-Based Meta Analysis (MBMA).
The predictive models created by Reveal Clinical solely belong to our life sciences partners. We are a software platform that enables the creation and utilization of predictive models. We do not use sponsor data to generate generalized models that are provided to other life sciences companies. Our platform does not learn from or train from any customer data.
Reveal Clinical uses a combination of internal trial data (preclinical or clinical) as well as external data sources (natural history data, external control arm trial data) and literature to build program and drug-specific predictive models.
Predictive models support activities across the life cycle of drug development. At the earliest stages, predictive models help teams assess the potential of preclinical candidates. At later stages, predictive models help teams identify the optimal patient population to reduce trial size and increase POS. As teams approach commercialization, predictive models help market access and commercial teams secure favorable pricing and formulary positioning.
Reveal Clinical has been built with the highest levels of data security and integrity standards. We align with key regulatory and compliance standards such as HIPAA, 21 CFR Part 11, GAMP5, and GDPR.
We'll walk through your disease area, your data, and how Reveal Clinical fits into your development timeline.